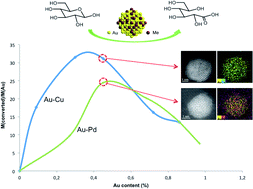
Supported bimetallic nanoparticles are particularly attractive catalysts due to increased activity and stability compared to their monometallic counterparts. In this work, gold-based catalysts have been studied as catalysts for the selective base-free oxidation of glucose. TiO2-supported Au–Pd and Au–Cu series prepared by the sol-immobilization and precipitation-reduction methods, respectively, showed a significant synergistic effect, particularly when the theoretical weight ratio of the two metals was close to 1 : 1 (with an actual experimental bulk Au/Pd molar ratio of ca. 0.8 and ca. 0.4 for Au/Cu) in both cases. XPS analysis showed that the presence of Auδ+, Pd2+ and CuOH species played an important role in the base-free glucose oxidation.
Au-based bimetallic catalysts: how the synergy between two metals affects their catalytic activity
RSC Advances, vol. 9, pg. 29888-29901 (2019)