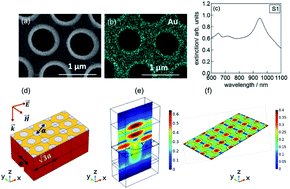
Plasmon-driven chemical transformation has become a promising approach for enhancing sluggish electrocatalytic reactions. Herein, an alternative enhancement strategy employing plasmon-induced hot electrons was developed and found to be competitive with oxygen reduction reaction (ORR) on platinum electrodes. We demonstrated that, by using the intertwined plasmon-catalytic-electrochemical properties of nanoperforated gold thin film electrodes, the ORR under alkaline conditions could be significantly enhanced by careful tuning of the laser wavelength and power density. Irradiation at 980 nm and 2 W cm−2 displays maximal current densities of j = −6.0 A cm−2 at 0.95 V vs. RHE, under hydrodynamic conditions, comparable to that of commercial Pt/C (40 wt%) catalysts, with good long-term stability. The wavelength-dependent electrochemical reduction confirmed that the hot carriers formed during plasmon decay are responsible for the improved electrocatalytic performance.
Plasmon-enhanced electrocatalytic oxygen reduction in alkaline media on gold nanohole electrodes
Journal of Materials Chemistry A, vol. 8, pg. 10395-10401 (2020)