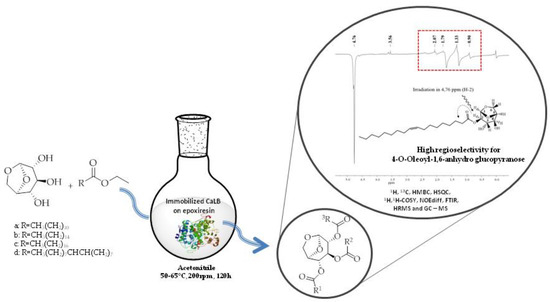
Every year, a large amount of residual agroindustrial waste has been generated and only around 10% is in fact reused. The development of new strategies for biomass valorization is important to add value to these commodities, since biomass is an excellent alternative feedstock to obtain chemicals of interest from renewable resources. The major compound of pyrolytic treatment of lignocellulosic biomass is levoglucosan (1,6-anhydroglucopyranose), an anhydro-sugar that can be transformed into glucose and is greatly valued in the most diverse industrial sectors as a surfactant, emulsifier, or even a lubricant. In this work, levoglucosan was acylated by lipase-catalyzed transesterification in acetonitrile with great conversions and selectivities with different acyl donors such as ethyl esters of lauric, palmitic, stearic, and oleic acids prepared in situ in an integrated strategy mediated by commercial lipases Novozym435 (N435), PSIM, and the home-made biocatalyst CaLB_epoxy. As a result, all biocatalyst generated mostly monoesters, with N435 being more selective to produce lauric esters (99% at 50°C) and PSIM to produce oleic esters (97% at 55 °C) while CaLB_epoxy was more selective to produce oleic esters of levoglucosan (83% at 55°C). This is the first report in the literature on the production of high selectivity levoglucosan esters
Regioselective Acylation of Levoglucosan Catalyzed by Candida Antarctica (CaLB) Lipase Immobilized on Epoxy Resin
Sustainability, vol. 11, pg. 6044 (2019)